Bronze disease is a type of corrosion process that affects bronze and other copper-based alloys. Not unlike usual bronze corrosion, where a patina starts to form on the surface due to oxidation of the copper, bronze disease is due to the process of oxidation, but is a rare and particularly damaging form of corrosion.
Chemical Process
Bronze disease is irreversible, and once the object has it, there is no treatment; there are only ways to slow it down. The chemical reaction is cyclic, meaning that it will just happen continuously unless there are no more reactants to react. The reaction occurs due to contamination of the object by saltwater or burial.
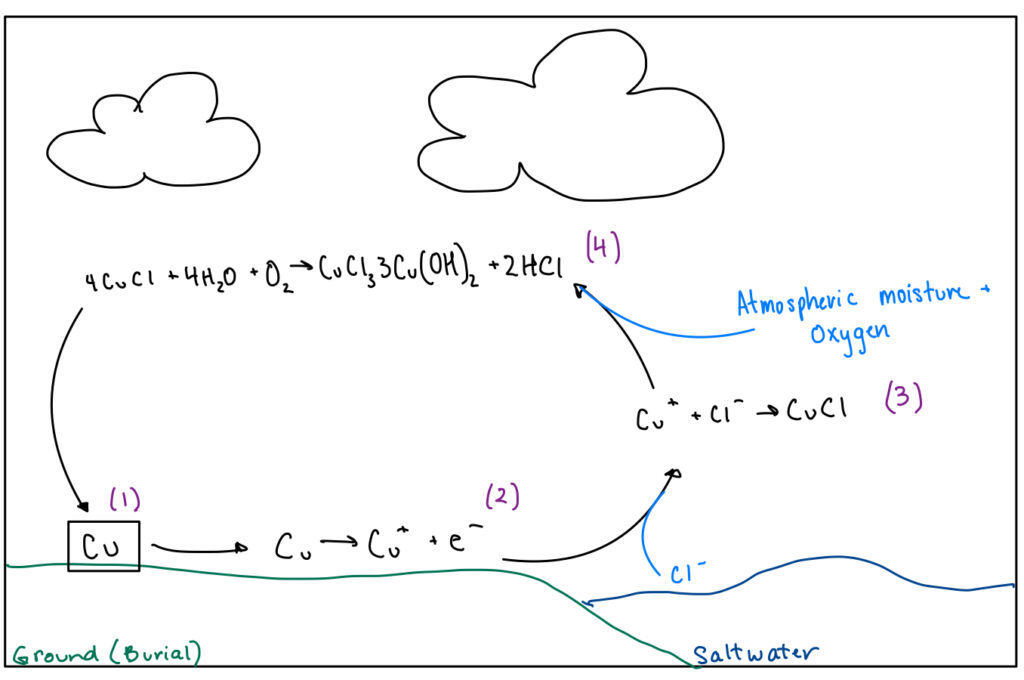
Due to electrochemistry and a high reduction potential, copper will oxidize to a cuprous ion when introduced to oxygen (2). This means that copper will lose an electron and become positively charged. This ion will react with the chloride ions around it. Chloride ions come from the dissociation of salt in saltwater, NaCl. Chloride ions are negative, and in an effort to have no charge, they will react with the positive cuprous ion to make cuprous chloride (3). This highly unstable salt, cuprous chloride, will continue to react with the atmospheric moisture and oxygen to form cupric chloride/cupric hydroxide compound and hydrochloric acid (4). This reaction is where the characteristic green bloom comes from and the pitting that eats away at the object. Due to excess copper, the object will continue to oxidize and react with the chlorides (1).
Characteristics of Bronze Disease
Bronze disease is visually characterized by a vibrant green coating. It can be seen as all shades of green and can look almost fuzzy due to the salts on the surface. Typically, bronze disease affects isolated patches of the object. A raised bloom of crystals and pitting into the object’s surface can be seen.
Based on these visual cues, objects were chosen. To the right, explore the visual differences seen between an object with no corrosion and one that possibly has bronze disease.


After: 1971.8.30 Roman- Bowl
Why does this matter?
When bronze disease was first identified, scientists thought it was an infection, contagious to all that is around it. Chlorides from a contaminated object can transfer and spread the condition through whole copper collections. Due to the nature of this ‘disease,’ there are only ways to slow down the process, which is removing it from moisture or oxygen to stop the cyclic reaction. Once a contaminated object is reintroduced to humidity and air, the cyclic reaction will continue right where it left off. Lack of information or proper analysis of these objects can lead to the destruction of complete objects.
One thought on “Materiality in Museums: Material Analysis of Bronze Disease”
Comments are closed.